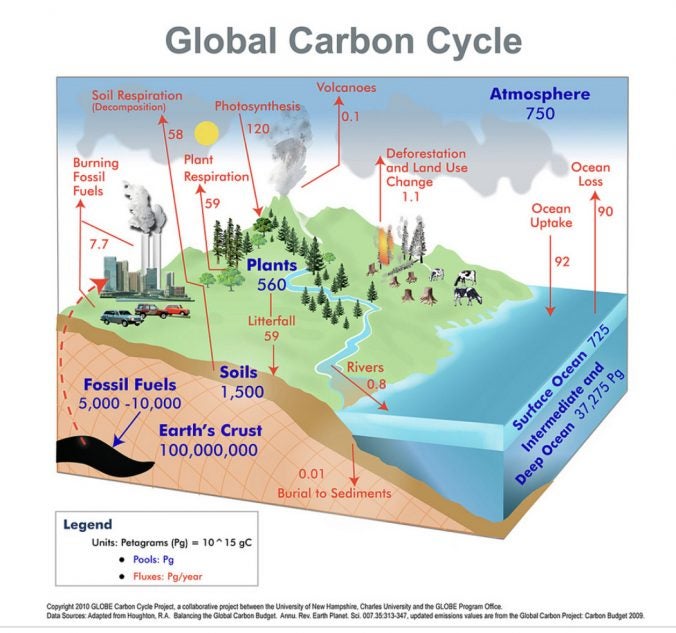
Most of the carbon in the ocean is in the form of bicarbonate ions, which can combine with seawater calcium to form calcium carbonate (CaCO3), a major component of marine organism shells.Carbon dioxide from the atmosphere dissolves in water, combining with water molecules to form carbonic acid, which then ionizes to carbonate and bicarbonate ions.The biogeochemical cycle occurs at a much slower rate than the biological cycle since carbon is stored in carbon reservoirs for long periods of time.The biological carbon cycle is the rapid exchange of carbon among living things autotrophs use carbon dioxide produced by heterotrophs to produce glucose and oxygen, which are then utilized by heterotrophs.Carbon is present in all organic molecules carbon compounds contain large amounts of energy, which humans use as fuel.Although much of the debate about the future effects of increasing atmospheric carbon on climate change focuses on fossils fuels, scientists take natural processes, such as volcanoes and respiration, into account as they model and predict the future impact of this increase. This is another example of how human activity indirectly affects biogeochemical cycles in a significant way. The large numbers of land animals raised to feed the earth’s growing population results in increased carbon dioxide levels in the atmosphere due to farming practices, respiration, and methane production. Carbon is released as carbon dioxide when a volcano erupts or from volcanic hydrothermal vents.Ĭarbon dioxide is also added to the atmosphere by the breeding and raising of livestock. Carbon sediments from the ocean floor are taken deep within the earth by the process of subduction: the movement of one tectonic plate beneath another. Another way for carbon to enter the atmosphere is from land by the eruption of volcanoes and other geothermal systems. A non-renewable resource is either regenerated very slowly or not at all. Fossil fuels are considered a non-renewable resource because their use far exceeds their rate of formation. Deeper underground, on land and at sea, are fossil fuels: the anaerobically-decomposed remains of plants that take millions of years to form. This carbon can be leached into the water reservoirs by surface runoff. On land, carbon is stored in soil as a result of the decomposition of living organisms or the weathering of terrestrial rock and minerals. Over geologic time, the calcium carbonate forms limestone, which comprises the largest carbon reservoir on earth. These organisms eventually form sediments on the ocean floor. Some of these ions combine with seawater calcium to form calcium carbonate (CaCO 3), a major component of marine organism shells. More than 90 percent of the carbon in the ocean is found as bicarbonate ions. \( \newcommand\): Formation of bicarbonate: Carbon dioxide reacts with water to form bicarbonate and carbonate ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
